Bistos BT350 Fetal Monitor
Offer end in:
Description
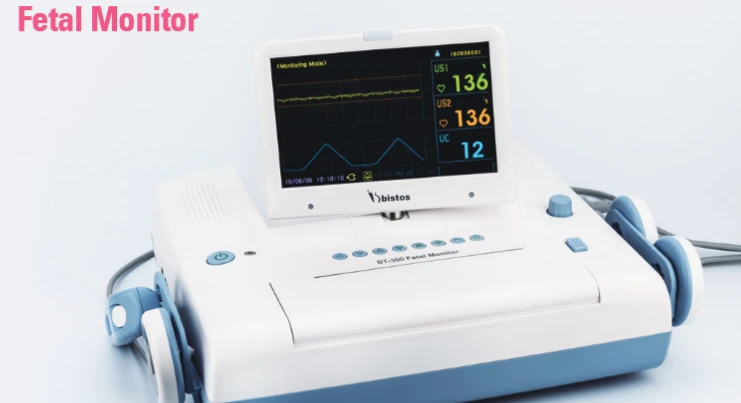
Bistos BT350 Fetal Monitor is a continuous fetal monitoring instrument manufactured in Korea by Bistos, designed for cardiotocographic assessment of fetal heart rate and uterine activity in maternity wards, labour rooms, obstetrics departments, and antenatal care settings. The instrument supports continuous monitoring mode and is equipped with waterproof probes that provide reliability and ease of cleaning during routine clinical obstetric monitoring use. The color LCD display with dimensions of 45mm x 25mm provides clear visualization of fetal heart rate trace and numerical data during active monitoring sessions, supporting efficient clinical observation by obstetric nursing and medical staff throughout the monitoring procedure. The instrument is supplied new and is manufactured in Korea, reflecting an internationally sourced product for the Indian hospital and maternity care market.
The fetal heart rate measuring range of 50 to 240 beats per minute supports comprehensive monitoring coverage across the clinically relevant fetal heart rate spectrum encountered during routine and high-risk obstetric monitoring procedures. The instrument incorporates alarm functions for bradycardia and tachycardia conditions, along with a signal loss alarm and sound alarm function, providing multi-parameter alert capability to support timely clinical response during active fetal monitoring sessions. The twin probe configuration supports simultaneous monitoring of twin pregnancies, and the instrument incorporates a prevention mechanism to reduce the risk of fetal heart rate miscalculation arising from twin fetus signal interference during monitoring, contributing to more reliable trace interpretation in multiple pregnancy monitoring scenarios.
The instrument features a built-in battery backup system that supports uninterrupted monitoring during power fluctuation or transition situations within clinical environments. A detachable thermal printer enables direct on-site printing of the cardiotocograph trace record, supporting paper documentation of fetal monitoring sessions within the clinical workflow. Stored data can be reviewed after monitoring sessions, with the instrument calculating and displaying parameters including baseline, acceleration, deceleration, and fetal movement data to support comprehensive trace analysis. Multilingual interface support across 13 languages including English, Chinese, Spanish, German, Russian, and Portuguese provides operational flexibility for diverse institutional environments. A built-in quick guide accessible through the menu screen supports staff orientation to instrument operation and maintenance procedures. Bistos BT350 Fetal Monitor is intended for use by trained obstetricians, midwives, and qualified obstetric nursing personnel only, in accordance with applicable fetal monitoring equipment operation and patient care guidelines.
Specifications of Bistos BT350 Fetal Monitor
| Shipping Weight | 7 kg |
| Shipping Dimensions | 40 × 35 × 30 cm |
| Monitoring Modes | Continuous |
| Thermal Printer | Detachable |
| FHR Analysis | Signal loss prevention, baseline, acceleration, deceleration, fetal movement |
| Probes | Waterproof twin probes |
| Color LCD | Yes LCD Display Size: 45 mm × 25 mm |
| FHR Measuring Range | 50–240 bpm |
| Multi-Language Support | 13 languages (English, Chinese, Spanish, German, Russian, Portuguese, etc.) |


 Medical Equipment
Medical Equipment
 Hospital Furniture
Hospital Furniture
 Medical Disposables
Medical Disposables
 Rehabilitation
Rehabilitation
 Diagnostic & Labware
Diagnostic & Labware
 Eyes & ENT
Eyes & ENT
 Critical Care Equipment
Critical Care Equipment
 Urology
Urology
 Aesthetic & Dermatology Equipment
Aesthetic & Dermatology Equipment
 Gastroenterology
Gastroenterology